Popular Science: First fluorescent dye for the imaging of amyloid fibrils
Amyloid fibrils are formed by the misfolding of specific proteins inside the cells. Even though their formation is linked to the development of neurodegenerative diseases, their role in the manifestation of these diseases remains unclear. Thus far, it has been possible to study these diseases using a range of imaging techniques (transmission electron microscopy, antibody staining). However, these techniques do not permit imaging of proteins within the living cells. Previously, researchers have attempted to solve the issue using fluorescent proteins fused with amyloid-forming proteins. However, this affects the fibril forming behaviour of the amyloids. To counter this problem, scientists have tried to use nanoparticles or small-molecule dyes but neither was successful.
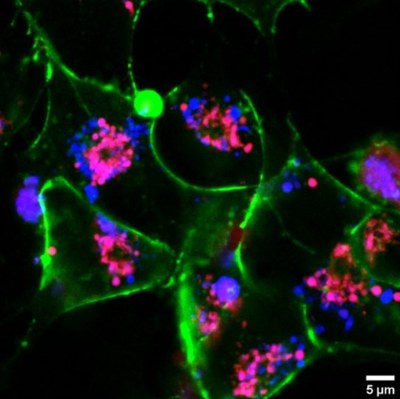 Human cells stained by RB1 (red) and amyloid-forming protein labelled with a dye (blue). Protein in a pathological fibril form is seen as magenta, the cell membrane as green.
Human cells stained by RB1 (red) and amyloid-forming protein labelled with a dye (blue). Protein in a pathological fibril form is seen as magenta, the cell membrane as green.source: SI of the original article
This is because a successful small dye molecule must exhibit several important properties, including 1) substantial selectivity for amyloid fibrils, 2) a substantial difference between fluorescence of the dye and dye bound to proteins, and 3) easy access to cells through cell membranes. Accordingly, RB1 and RB2 dyes have been developed. RB1 displays exceptional fluorescence in the presence of amyloid fibrils due to its high affinity with the amyloids. This can be explained by the shape of the RB1 molecule, which fits perfectly into the hydrophobic pocket of fibrils. This truly exceptional affinity has led scientists to believe that the RB1 molecule will selectively bind to amyloid fibrils, even in a complex cellular environment. This hypothesis has been tested on two human cell lines and was confirmed in both experiments. The dye selectively binds to amyloid fibrils and hence allows high contrast imaging of their formation in living cells.
In addition to the expected effect of dying cytosolic fibrils, the RB1 molecule can also dye nucleoli. The nucleolus is morphologically distinct from the fibrils so the staining of nucleoli can be used as an internal control for the performance of the dye. Moreover, fibrils are never found inside the cell nucleus.
A necessary requirement for any substance used in living cells is low or absent toxicity. Scientists therefore tested the toxicity of RB1 on both cell lines and found that cell viability was largely unaffected by the application of the dye.
In conclusion, Czech scientists have managed to develop the very first fluorescent dye to allow selective imaging of amyloid fibrils in cytoplasm of living cells. This discovery could be utilised to study the mechanism underpinning Parkinson’s and other neurodegenerative diseases.
Magda Křelinová
Pankaj Gaur, Maksym Galkin, Andrii Kurochka, Subrata Ghosh, Dmytro A. Yushchenko, and Volodymyr V. Shvadchak; ACS Chemical Neuroscience 2021 12(8), 1293–1298
Document Actions